Flow of heat from a cold body to a hotter body. So, this type of process is non-spontaneous at all temperature.
#Spontaneous processes free#
Spontaneous and non-spontaneous refer to ΔG, or the free energy change of the reaction. By my understanding, that's all that is meant by "non-spontaneous." Below the boiling point, the process in non-spontaneous as it requires energy in form of heat for the process to occur. The entropy change for the process \ is 22.1 J/K and requires that the surroundings transfer 6.00 kJ of heat to the system. Spontaneous processes often require activation energy, but do not require a prolonged input of energy.
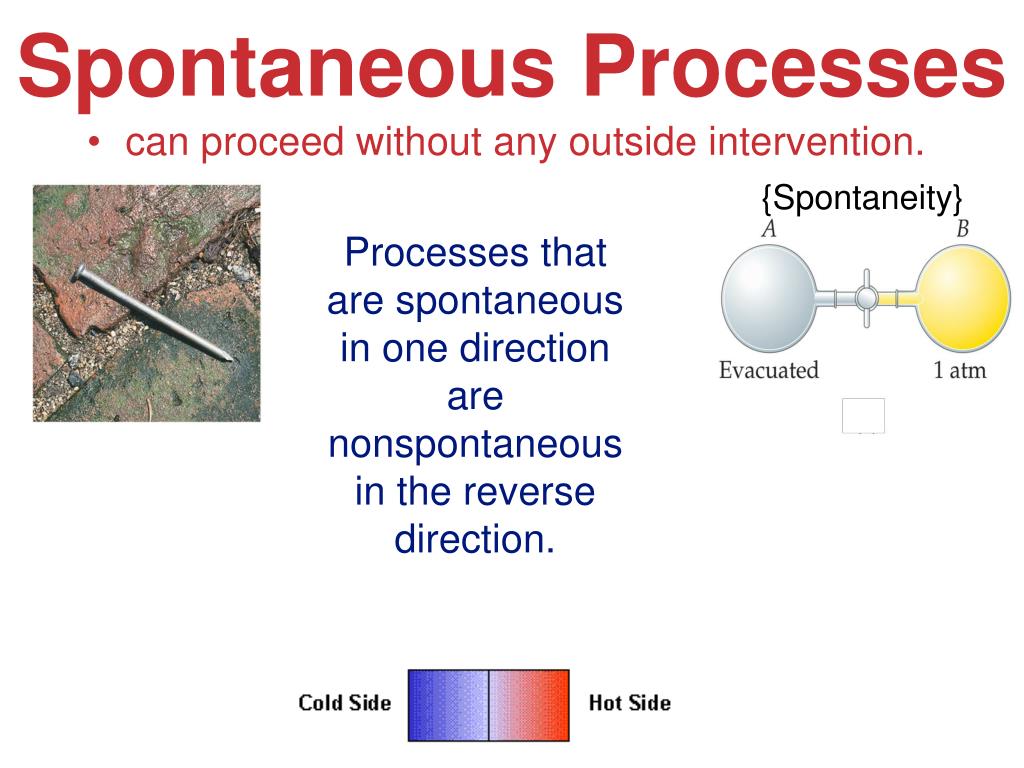
A spontaneous process is one that occurs naturally under certain conditions. Which of the following processes is a non-spontaneous process? Drying of leaves, spoilage of food, cooling of water and waterfall (assuming water falling) are all spontaneous processes since they all proceed without application of external force. To get the reverse process to occur, the water temperature must be lowered to 0☌. An exothermic reaction may be non-spontaneous if the reaction also leads to a decrease in entropy. A process which can neither take place by itself nor by initiation is called a non spontaneous process. 3) Diffusion of gas from low pressure to a high pressure. What is spontaneous and non spontaneous process? Systems undergoing a spontaneous process may or may not experience a gain or loss of energy, but they will experience a change in the way matter and/or energy is distributed within the system.

2) Flow of heat from cold body to a hot body. In other words, the process would not happen on its own, as … Examples are the reverse of spontaneous processes: water does not flow from a lower level to a higher level heat does not flow from a cold body to a hot body solute particles in a solution do not gather together in one part of the solution etc. positive, the process is non-spontaneous as written, but it may proceed spontaneously in the reverse direction.

We can divide these reactions into two categories as spontaneous … If there's an endothermic reaction that is spontaneous, then the reverse reaction is an exothermic reaction that's non-spontaneous.
